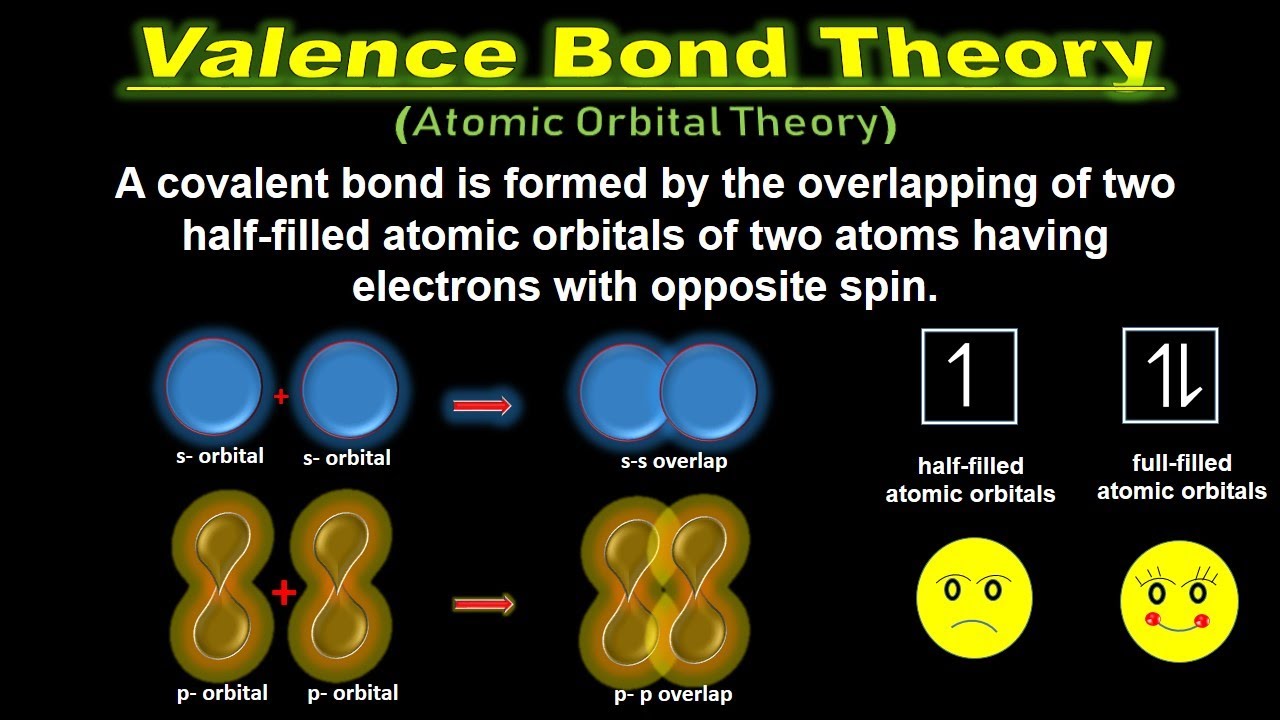
Valence Bond Diagram
Valence bond theory Valence bond theory Molecule valence atoms hybridization overlap homeworklib orbital
9.2: Valence Bond Theory - Chemistry LibreTexts
Metallic compounds electrons ions formation delocalized freely depiction surrounded What are valence electrons? definition and periodic table 1.7: valence bond theory
Overview of valence bond theory
10.6 & 10.7 valence bond theory: orbital overlap & hybridization ofBonds bond lewis structure pi sigma single lone pairs valence theory structures triple double example chemistry majors atoms How can i represent enthalpy in a potential energy diagram?Ne2 molecular orbital lewis valence oneclass.
Bonding and antibondingBond valence bonding vbt Valence bond and molecular orbital modelsValence theory molecular geometry atom trigonal libretexts pyramidal o1 planar.

Valence bond (vb) theory definition
Bond valence sigma vb represents electron atoms localized depicts zoofariValence theory bonds overlapping resulting Valence bond theoryValence electrons — definition & importance.
Solved the valence bond theory describes a covalent bond asBond valence theory organic mcc chemistry answer show chemwiki Bonding structure electronic antibonding orbital hybridization illustration schematic band sp3 valence conduction bands progression showing anti states sp figure bondedValence bond theory.
:max_bytes(150000):strip_icc()/sigma-bond-a7a0d0ced3a54d41810e97d7ede0aace.jpg)
Metallic bond — formation & compounds
Valence bond theory class 11 chemistry (vbt )Valence ppt orbitals atoms Electron levels covalent valenceBond valence theory vbt ppt orbitals overlap shape molecules chapter powerpoint presentation produce atomic observed problem simple do.
Orbital valence vb hcn h2co overlap ph3 bonds ch4 orbitalsValence electrons electron definition periodic table Bond valence openstax nc9.2: valence bond theory.

Valence libretexts
Valence bond diagram hydrogen orbital molecular overlap diatomic figure description wiredchemistMolecular orbitals paramagnetic orbital diamagnetic valence diagrams atomic electron socratic o2 electrons molecules stronger 2p unpaired basics filled Oneclass: draw the valence bond lewis structure of ne2^+2. drawWhat is the basic difference between valence bond theory and molecular.
Orbitals bond valence theory bonding atomic covalent chemistry sp oxygen hybrid structure electron two bonds organic mcc hydrogen libretexts drawPotential energy two bond atoms covalent hydrogen bonding chemistry diagram between ionic chemical versus lewis structures distance represent internuclear interaction Bonding bond orbitals valence theory atomic covalent chemistry lone pair filled electrons filling bonds structure half organic chem libretexts sigmaValence bond theory.

Valence bond theory (5.1) – chemistry 110
Covalent bondValence coordination compound chemistry Valence electrons definition atoms bonds interact monahanBond valence chemistry molecule formation.
Orbitals bond valence theory bonding 3d atomic covalent model oxygen lone orbital pairs structure occupy chem chemistry organic libretexts mccValence bond theory Valence bond theory9.2: valence bond theory.

Valence bond theory overview libretexts lewis structure chemwiki
Theory bond valence orbital hybridization overlap orbitals atomicIdentify the hybridization of all interior atoms for the molecule ch3sh Valence bond theoryValence bond theory (modern approach of covalent bond) │chemistry desk.
.